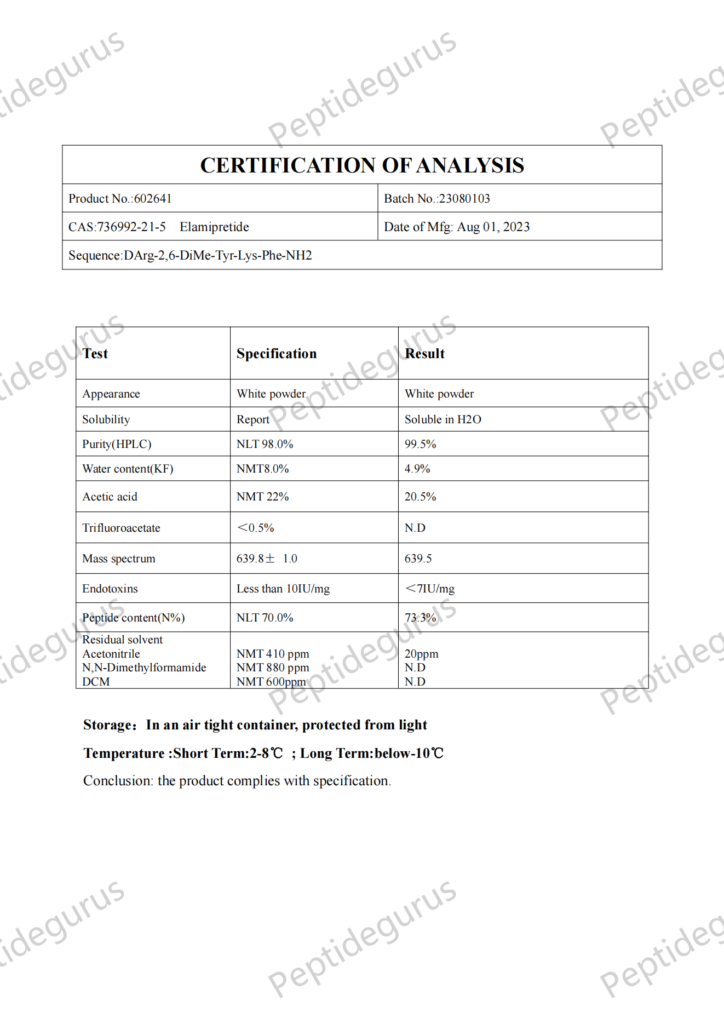
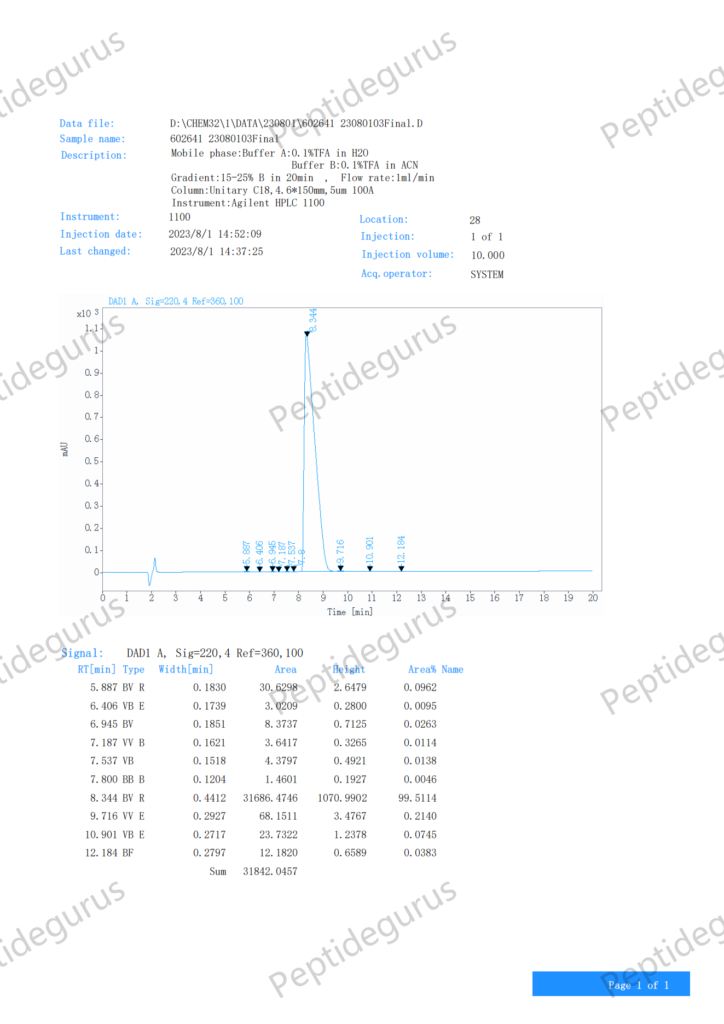
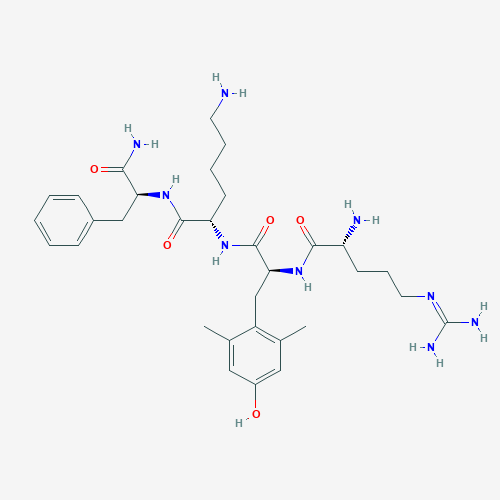
Cos'è SS-31?
SS-31 (elamipretide) is a small, aromatic peptide that easily penetrates cell and organelle membranes.It is thought to interfere with the production of reactive oxygen species (ROS or free radicals) and promote energy production in cells by stabilizing the enzyme cardiolipin within mitochondria. Cardiolipin is part of the inner mitochondrial membrane where it acts as a fundamental component of the electron transport chain, the machinery by which most energy need for cellular functioning is produced.
Dysfunction of cardiolipin has been implicated as contributing to the pathology of a number of diseases including Alzheimer’s disease, Parkinson’s disease, nonalcoholic fatty liver disease, diabetes, heart failure, HIV, cancer, chronic fatigue syndrome, and more. Cardiolipin is thought to be a major component of mitochondrial myopathy, which isn’t a single disease but rather is a group of neuromuscular disorders caused by damage to mitochondria. Mitochondrial myopathy is characterized by everything from muscle weakness and exercise intolerance to heart failure, seizures, and dementia. SS-31 is the first peptide to ever undergo clinical trials as a potential treatment for mitochondrial myopathy.
Miglioramento dei mitocondri
Primary mitochondrial diseases (PMDs) are among the most common inherited conditions in the world. They are caused by dysfunction in the energy-producing apparatus of mitochondria. Symptoms vary greatly between forms of the disease, but the most susceptible organ systems are those with high energy demands (e.g. nervous system, heart, kidney, etc.). Muscle involvement and exercise intolerance are almost universal in mitochondrial disorders. Common symptoms include easy fatigue, exercise intolerance, and seizures.
PMDs, and mitochondrial diseases in general, are primarily characterized by disturbances in the production of ATP. ATP acts as the energy currency of the cell and is necessary to almost every cell function. Stabilizing ATP production in the setting of mitochondrial disease has long been a goal of the medical profession. With the development of SS-31, that goal may final have been realized.
The first evidence that SS-31 could restore energy production in PMDs came from animal studies. In that research, rats who had suffered ischemia-perfusion injury (a non-genetic cause of mitochondrial disease) of the kidney were given SS-31. The peptide protected kidney structure, accelerated recovery of ATP production, and reduced cell death and necrosis within the kidney[1]. Subsequent studies in mice showed that SS-31 interacted with cardiolipin in the inner mitochondria membrane and revealed that the peptide could reduce symptoms of mitochondrial disease regardless of etiology. There is also evidence that it can improve mitochondrial dysfunction that results from age[2]–[4]. From these findings, it was relatively simple to convince the FDA to grant orphan drug status to SS-31 and pave the way to clinical trials.
In phase II trials in humans, SS-31 increased exercise performance after just 5 days of treatment and showed no safety concerns or prominent side effects[5]. Unfortunately, phase III trials failed to produce convincing evidence of SS-31’s clinical utility[6]. That said, there is good reason to believe that the trial endpoints simply weren’t appropriate and that additional work will result in the peptide being approved for the treatment of certain mitochondrial conditions. According to Dr. Bruce Cohen, director of the Neurodevelopment Science Center at Akron Children’s Hospital, the results for prior phase II clinical trials were very encouraging and so it is not time to give up. Rather, he notes, SS-31 should spur interest in this particular area and bring other big pharma research to the table[7]. It appears that that is already happening as the company that first brought SS-31 to clinical trials is planning to move forward with trials of a derivative of SS-31 as well as trials investigating other endpoints for SS-31 treatment[6].
As of right now, SS-31 is being tested in a number of different human diseases and under a number of different trial models. The peptide is considered safe to use in humans, so it can also be prescribed by doctors under compassionate care exceptions to patients who have no other treatment options. The peptide will likely become part of mainstream medical care for a number of conditions in the near future, but even now it is available to people who need it while clinical trial work is ongoing.Ischemia
Perhaps the most compelling secondary application of SS-31 is in the treatment of heart failure. It has long been known that heart failure causes negative changes to the function of mitochondria and that these changes, in a kind of destructive cycle, cause heart failure to worsen. Research in human heart tissue treated with SS-31 shows significant improvements in mitochondrial oxygen flux and activity of specific components involved in the production of ATP. This particular study was carried out in a manner that precluded cardiolipin restructuring, however, suggesting that SS-31 may have a second mechanism of action on mitochondrial function that needs to be explored[4]. This finding has actually been replicated in a number of research studies, strengthening the idea that SS-31 is not just useful for restoring ATP production via cardiolipin interaction. The peptide is actively being investigated for its ability to alter the production of reactive oxygen species and improve mitochondrial function in both acute and chronic use situations.
Studies in dogs, for instance, show that chronic treatment with SS-31 can improve left ventricular function in the setting of advanced heart failure. Measures of mitochondrial respiration and maximum ATP synthesis correlated well in this study with overall improvement in left ventricular function implying that SS-31 could be an effective long-term treatment for improving energy dynamics and reducing cardiac remodeling in advanced heart failure[8].
Trials exploring the use of SS-31 in ST-segment elevation myocardial infarction (heart attack) found that the peptide can drastically reduce levels of HtrA2. HtrA2 is a measure of cardiomyocyte apoptosis. These results suggest that SS-31 may be useful in the context of acute heart attack to reduce the extent of injury and preserve cardiac tissue[9].
One role of mitochondrial-targeted therapy in heart failure: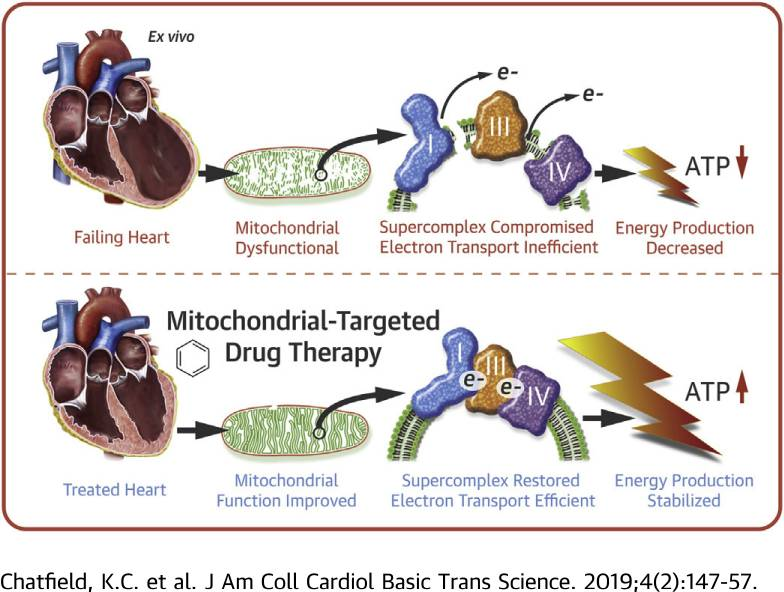 Fonte:Pubch
Fonte:Pubch
Diabete
Il diabete, sebbene apparentemente causato da una semplice inadeguatezza nella secrezione o nella funzione dell'insulina, è una condizione complessa con molteplici manifestazioni patofisiologiche. Negli ultimi anni, c'è stato un crescente interesse nel ruolo della compromissione mitocondriale nella patogenesi della malattia, in particolare nel diabete di tipo 2. Il trattamento della disfunzione mitocondriale sarebbe quindi un modo per migliorare alcune delle conseguenze a lungo termine del diabete come il danno ossidativo a piccoli vasi. In uno studio sull'uomo somministrato SS-31, è stata osservata una marcata riduzione della produzione di specie reattive di ossigeno. Ciò suggerisce che SS-31 può aiutare a ridurre il danno ossidativo che di solito accompagna la disfunzione mitocondriale e può quindi rallentare o fermare la progressione della malattia microvascolare nel diabete di tipo 2. Questa ipotesi è ulteriormente confermata dalla scoperta, nello stesso studio, che SS-31 ha aumentato i livelli di SIRT1. I livelli di SIRT1 sono stati associati a una migliore sensibilità all'insulina e alla riduzione dell'infiammazione nel diabete di tipo 2 [10].Riduce l'infiammazione
A theme throughout the sections above is inflammation and the ability of SS-31 to reduce it. In particular, SS-31 appears to be a potent regulator of reactive oxygen species (free radicals) and thus helps to reduce the serious oxidative stress that arises from long-term illness such as diabetes, heart disease, and more. Research in cell cultures suggests that SS-31 reduces inflammation and oxidative stress by reducing expression of FIS1[11]. FIS1 is a mitochondrial protein that is important for mitochondrial growth and division. Elevated levels of FIS1 have been observed in a number of neurodegenerative diseases as well as a variety of cancers and are thought to be evidence of dysfunctional mitochondrial division secondary to dysfunction and inflammation.
There is also good evidence from mouse models to show that SS-31 reduces levels of the inflammatory cytokine CD-36, reduces expression of activated MnSOD, suppresses NADPH oxidase function, and inhibits NF-kappaB p65[12]. All of these are markers of high oxidative stress, so reducing their levels is indicative of reduced free radical production and an improved inflammatory status in the cell. NF-kappaB expression, in particular, is heavily associated with cellular inflammation and is chronically active in a number of inflammatory diseases like rheumatoid arthritis and inflammatory bowel disease. With SS-31, mitochondria do not undergo inflammasome activation, which is to say they don’t convert from the primary production of ATP to primarily producing ROS.
Inflammasome activation is avoided and normal mitochondrial function is preserved in the setting of SS-31 administration: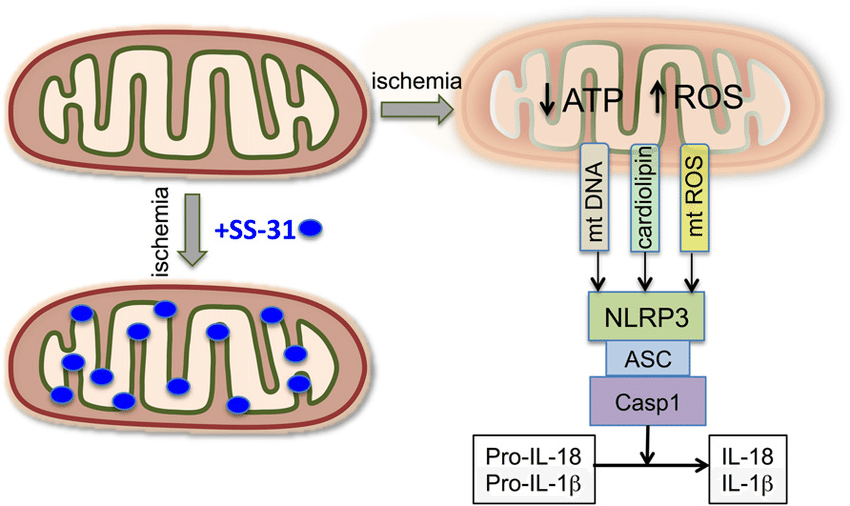 Fonte:ResearchGate
Fonte:ResearchGate
Riepilogo SS-31
Sebbene SS-31 fosse originariamente di interesse perché si pensa che regoli la funzione mitocondriale nel contesto della malattia mitocondriale, ci sono anche buone prove che il peptide possa regolare l'infiammazione indotta dai mitocondri. C'è molto interesse attivo nell'uso di SS-31 per migliorare la funzione mitocondriale e quindi la produzione complessiva di energia tramite sintesi di ATP. Sebbene gli studi iniziali di fase III non abbiano avuto successo, si pensa che questo possa essere più il risultato degli endpoint misurati rispetto a un vero fallimento del peptide ad avere alcun effetto. Attualmente sono in corso studi di fase II e studi di fase III pianificati per testare SS-31 in una varietà di stati patologici diversi e con una varietà di diverse misure di esito. SS-31 può benissimo fornire la chiave per comprendere la disfunzione mitocondriale in una varietà di malattie e quindi può rivelarsi utile nella progettazione di trattamenti avanzati per la malattia di Alzheimer, la malattia di Parkinson, le malattie cardiache, il diabete, le malattie renali e altro ancora.Autore dell'articolo
La letteratura di cui sopra è stata studiata, modificata e organizzata dal Dr. E. Logan, M.D. Dr. E. Logan ha conseguito una laurea in dottoratoCase Western Reserve University School of Medicinee un B.S. in biologia molecolare.Autore della rivista scientifica
Nel suo lavoro per scoprire trattamenti per i pazienti con malattie mitocondriali, il dott. Cohen è diventato uno dei principali esperti del mondo. Sebbene non ci siano cure per le malattie mitocondriali, il Dr. Cohen ne cerca ogni giorno. Come risultato del suo lavoro e dell'impegno del nostro ospedale ad aiutarlo a costruire un'infrastruttura per condurre studi clinici mitocondriali, Akron Children è una delle principali istituzioni del paese che ricercano trattamenti. Prima di unirsi a Akron Children's, il Dr. Cohen è stato capo della neurologia pediatrica per la clinica di Cleveland. Ha conseguito la laurea in medicina presso l'Albert Einstein College of Medicine e ha completato la sua residenza pediatrica presso l'ospedale pediatrico di Filadelfia, la residenza di neurologia pediatrica presso il Columbia Presbyterian Medical Center e la sua American Cancer Society Neuro-oncology Fellowship a Philadelphia.Bruce H. Cohen, M.D.viene citato come uno dei principali scienziati coinvolti nella ricerca e nello sviluppo di SS-31. Questo medico/scienziato che approva o sostiene l'acquisto, la vendita o l'uso di questo prodotto per qualsiasi motivo. Non c'è affiliazione o relazione, implicita o altro, tra
Guru peptidicie questo dottore. Lo scopo di citare il medico è di riconoscere, riconoscere e accreditare gli sforzi esaustivi di ricerca e sviluppo condotti dagli scienziati che studiano questo peptide. Dr.Cohen è elencato in [5] sotto le citazioni di riferimento.